Peat bogs are quilted wetlands in which spongy masses of mosses such as sphagnum mosses are soaked with water and generate an acidic environment that is very limiting for the development of many micro-organisms, but which is paradoxically extraordinarily rich in biodiversity.
It is estimated that 3% of the emerged territories correspond to these wetlands, which fix in the substrate up to 30% of all the CO2 incorporated by living beings into the biogeochemical carbon cycle.
The peatlands of the southern hemisphere, such as those in the southernmost lands of the American continent in Chile and Argentina, have very marked singularities that differentiate them from others scattered around our planet and those found in Iberian territory. In addition to their considerable extension, their acidity is very pronounced, and this fact seems to limit the growth of algae such as desmids and diatoms.
Despite these extreme conditions, these ecosystems are an ideal refuge for many species of thecate amoebae (shelled amoebae) which find their paradise here, with hundreds of forms and species - many of them still to be discovered - and which play an important role in the renewal and balance of the peatlands.
Recently, thanks to the invitation of the University of La Frontera in Chile to give the first international course on peatland microscopy in South America in the laboratories of the Faculty of Agricultural Sciences and Environment of this University in the city of Temuco, we were able to test an equipment provided by Motic Europe: a Motic Panthera C2 trinocular microscope equipped with an epifluorescence module and with condenser and objectives to be able to carry out observations also with phase contrast, darkfield and polarised light techniques.
Within this framework of collaboration between Motic Europe, the Duero-Douro International Biological Station and the University of Temucco, we are carrying out research work on the samples collected in the Quillaipe peat bogs by Professor Rubén Carrillo, Head of the Department of Agronomic Sciences and Natural Resources and his team, trying to identify and study the microorganisms that inhabit these enclaves.
For this, a fundamental work is the live microscopic observation of the material collected using different lighting techniques, and the photographic record of the organisms that we find.
As a small initial sample of this work, we show here some microphotographs of the leaves of some of the most common sphagnums, in this case Sphagnum magellanicum together with several shots taken of some of the pleasant thecadas that live among these mosses: Nebela and Euglypha.
The Nebela species shown here, photographed with three different techniques: phase contrast, darkfield, and epifluorescence, is very similar to Nebela tincta, but also bears many similarities to all of this recently studied group. mainly by Kosyakan, Lara and other authors, who are trying to find the relationship between morphological and genetic diversity within the Nebela tincta-collaris-bohemica species complex, and other taxa such as Nebela gimlii.
Nebela gimlii. 400X. Phase contrast.
Nebela gimlii. 400X. Darkfield.
Nebela gimlii. 400X. Epifluorescence and Darkfield.
Almost all of them, studied and found in the northern hemisphere, are very similar morphologically, and apparently also have a very close relationship with the one found in the Quillaipe bogs that we show here and that could be a species very close to Nebela gimlii not yet described.
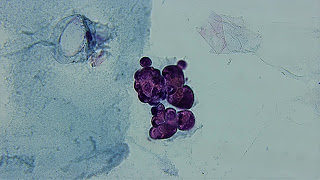
On the other hand, the species of Euglypha that we have photographed with the phase contrast technique, probably Euglypha ciliata whose contours resemble those of a light bulb, has its theca flattened and is covered with oval silica flakes that when superimposed seem to form a hexagonal mesh and as a particular feature it has fine siliceous spines on some of its plates.
Both amoebae, which are very voracious, live protected inside this shell-like structure, in which they live as if it were their home.
Euglypha ciliata. 400X. Phase contrast
Euglypha ciliata. 400X. Phase contrast
The cell walls of the sphagnum cells have a great hygroscopic capacity and can retain water between the empty spaces forming this closed reticular lattice that can be seen in the images and which is shown photographed at different magnifications with darkfield, phase contrast and polarised light techniques using the Motic Panthera C2 trinocular microscope at x100, x200 and x400 using different illumination techniques: Brightfield, darkfield, phase contrast and epifluorescence, using the FLED module and Motic dry objectives x10//PH1, x20/0. 45/PH1 and x40 /0. 65/PH2 (WD 0.6mm).
Leaf tissue of the moss (Sphagnum). 400X. Darkfield.
Leaf tissue of the moss (Sphagnum). 200X. Darkfield and Polarization.
© Antonio Guillén Oterino

























%20.%20Campo%20oscuro%20%20x%20400.jpg)
%20.%20Campo%20oscuro%20y%20polarizaci%C2%A2n%20%20x%20200.jpg)